Prescription drugs and pharmaceuticals are often at the center of many lawsuits. Manufacturers must undergo appropriate tests to meet the criteria set by the U.S. Food and Drug Administration (FDA) before releasing a product to the market. When the FDA properly licenses a product, it does not affect the manufacturer’s liability for someone injured due to medication defects.
If a prescription drug or medication has caused you illness or injury, you have the right to seek compensation and take legal action against all involved parties. This falls under a product liability lawsuit. You need not prove anyone’s negligence; instead, you must demonstrate the product caused harm while using it as intended without alteration.
. If a defective medication has harmed you or a loved one, Spector Injury & Accident Lawyers’ expert attorneys are explaining the various categories under which you can file your bad drug liability case and how to pursue proper compensation.
DESIGN DEFECTS
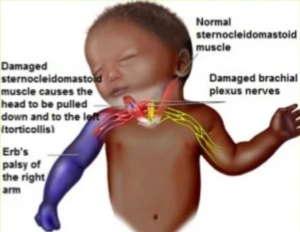
Design defects categorize a flaw in product design. The FDA may approve a medication, but it may lead to unforeseen side effects post-market distribution, posing serious harm to individuals. In such claims, the product’s manufacturing aligns with FDA standards, yet the design triggers dangerous side effects.
MANUFACTURING DEFECTS
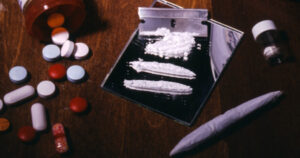
Manufacturers create defects during the manufacturing process by using unsafe or tainted ingredients. They may also mislabel the medication.
MARKETING DEFECTS
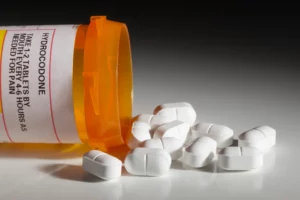
Marketing defects, also called failure to warn, occur when the manufacturer doesn’t provide the doctor or patient with any known drug interactions or side effects. The FDA mandates manufacturers to furnish this information, and withholding it can lead to the manufacturer’s responsibility for any resulting injuries or illnesses. Doctors and physicians can also face accountability if they neglect to provide this information.
SPEAK TO AN EXPERIENCED PERSONAL INJURY LAWYER
Even though medications are meant to ease, treat, and prevent illnesses, you have the right to receive compensation when a bad drug seriously harms you. Your personal injury lawyer can help you file the correct claim and identify all the appropriate parties involved in your lawsuit. If you feel you are the victim of a product liability case in the form of a bad or dangerous drug defect, don’t bear the weight of the case alone—let the qualified team at Spector Injury & Accident Lawyers help you! A Baltimore bad drug attorney will fight with you to ensure you get the compensation you deserve and protect your rights. Don’t hesitate—contact us today!
Spector Law Group